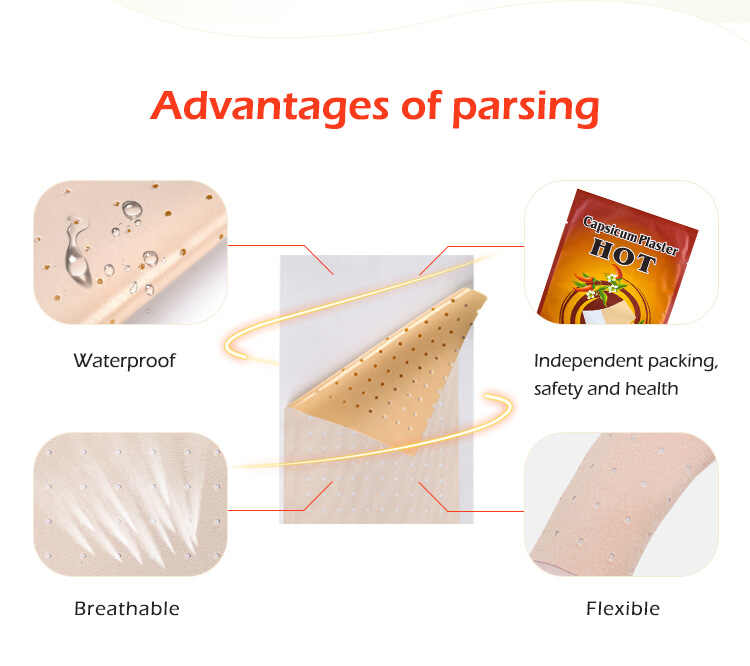
FDA, CE, ISO Certifications: Why They Matter for Patch OEM Buyers
The Short Answer
FDA, CE, and ISO certifications matter because they ensure your transdermal patch products are safe, compliant, and legally accepted in international markets, helping you avoid regulatory risks and build long-term brand trust.
In Simple Terms
If you're sourcing from a transdermal patch OEM manufacturer, certifications are not optional—they are your gateway to global distribution and a key factor in whether your products can be sold on platforms like Amazon or enter regulated markets like the U.S. and Europe.
1. What Is FDA Certification and Why It Matters
The U.S. Food and Drug Administration (FDA) regulates products entering the United States market.
Key Points:
Ensures product safety and manufacturing standards
Requires facility registration for many patch manufacturers
May involve OTC drug compliance depending on product claims
Why It Matters for Buyers:
Without FDA-related compliance, your products may face:
Customs rejection
Amazon listing removal
Legal liability risks
? For any brand targeting the U.S., FDA compliance is essential.
2. What Is CE Marking and Its Importance
The CE marking indicates that a product meets European Union safety, health, and environmental requirements.
Key Points:
Required for certain product categories in Europe
Demonstrates conformity with EU regulations
Increases buyer confidence
Why It Matters:
If your patch products fall under regulated categories in Europe, CE marking is necessary to:
Enter EU markets legally
Avoid import restrictions
Work with European distributors
3. What Is ISO Certification in Patch Manufacturing
The International Organization for Standardization (ISO) sets global standards for quality management systems.
Common ISO Standards:
ISO 9001 (Quality Management)
ISO 13485 (Medical Device Quality Management)
Why It Matters:
ISO-certified manufacturers:
Follow strict production processes
Maintain consistent product quality
Reduce defect rates
? This is especially important for private label transdermal patch buyers who rely on stable quality.
4. The Risks of Working with Non-Certified Manufacturers
Choosing a supplier without proper certifications can lead to serious business risks.
Common Problems:
Inconsistent product quality
Regulatory violations
Shipment delays or rejections
Platform bans (Amazon, Google Ads, etc.)
Real Impact:
A low-cost supplier without certifications may cost you far more in lost time, reputation, and compliance issues.
5. How Certifications Build Trust and Brand Value
Certifications are not just regulatory requirements—they are also powerful marketing assets.
Benefits:
Increase consumer confidence
Improve conversion rates
Support premium pricing strategy
Strengthen distributor relationships
? In competitive markets, certified products stand out.
6. What Patch OEM Buyers Should Verify
Before choosing a transdermal patch manufacturer, buyers should verify certification authenticity.
Checklist:
Valid FDA facility registration
CE documentation (if applicable)
ISO certificates with issuing body
GMP-compliant production facility
Pro Tip:
Always request:
Certification copies
Audit reports
Product testing data
7. Certifications + OEM = Scalable Global Business
A professional transdermal patch OEM manufacturer with FDA, CE, and ISO certifications enables:
Faster market entry
Lower compliance risks
Easier expansion into new regions
? This combination is critical for brands aiming to scale internationally.
Key Takeaways
FDA, CE, and ISO certifications are essential for patch OEM buyers because they:
Ensure product safety and compliance
Enable access to global markets
Reduce regulatory and business risks
Enhance brand credibility and trust
? Choosing a certified manufacturer is one of the most important decisions for long-term success.
Frequently Asked Questions (FAQ)
1. Do all transdermal patches require FDA approval?
Not all patches require full FDA approval, but most require FDA facility registration and compliance, especially for the U.S. market.
2. Is CE certification mandatory for all patch products?
CE marking is required if the product is classified under applicable EU regulations, particularly for medical-related uses.
3. What is the most important ISO standard for patch manufacturers?
ISO 9001 is essential for quality management, while ISO 13485 is important for medical-grade products.
4. Can I sell patches without certifications?
In some markets, yes—but it significantly increases risks and limits your ability to scale globally.
5. How can I verify a manufacturer’s certifications?
Request official documents and verify them through issuing organizations or third-party audits.
Ready to Work with a Certified Patch OEM Manufacturer?
If you're looking for a reliable, certified transdermal patch OEM partner, we offer:
✅ FDA-registered facility
✅ CE-compliant products
✅ ISO-certified quality management
✅ GMP production environment
? Contact us today for a free consultation and product samples.
? Build your brand with a trusted, globally compliant patch manufacturer.