How to Validate Your Transdermal Patch Idea Before Production
Introduction
Launching a transdermal patch product requires careful validation before committing to full-scale production. Patch product validation is a critical process that helps entrepreneurs and businesses minimize risks, optimize formulations, and ensure market success. This comprehensive guide explores proven methods for transdermal patch market test and patch sample testing to validate your product concept effectively.
Why Validation is Essential Before Production
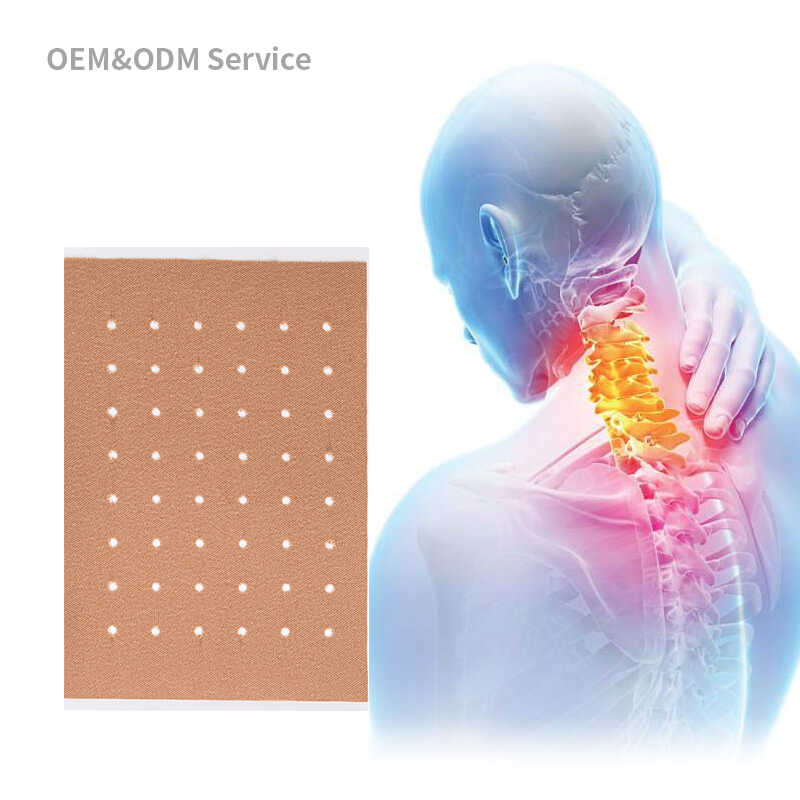
Investing in transdermal patch production without proper validation can lead to significant financial losses and market failures. A comprehensive patch product validation process helps you:
- Identify Formulation Issues - Discover adhesion, skin compatibility, or efficacy problems early
- Reduce Financial Risk - Avoid large upfront investments in products that may not sell
- Refine Market Positioning - Test different claims and target audiences before launch
- Gather Customer Feedback - Understand real user needs and preferences
- Optimize Pricing Strategy - Validate price points based on perceived value
Partnering with an experienced OEM patch manufacturer that offers sample development ensures your validation process produces actionable insights.
Step 1: Define Your Product Concept
Before conducting any patch sample testing, clearly define your product concept:
- Target Condition - What specific pain or health issue will the patch address?
- Active Ingredients - Which APIs or natural ingredients will be included?
- Unique Value Proposition - What differentiates your patch from competitors?
- Target Market - Who are your ideal customers (age, lifestyle, income level)?
- Price Point - What price range aligns with your positioning and margins?
Step 2: Conduct Transdermal Patch Market Test
Market Research Methods
Effective transdermal patch market test strategies include:
- Online Surveys - Use platforms like SurveyMonkey or Google Forms to gather quantitative data
- Focus Groups - Conduct in-person or virtual sessions with potential customers
- Social Media Polls - Test concepts on Instagram, Facebook, or LinkedIn communities
- Competitor Analysis - Review customer reviews and feedback on similar products
- Landing Page Testing - Create pre-launch pages to measure interest and collect leads
Key Metrics to Measure
- Purchase Intent - Percentage of respondents likely to buy
- Willingness to Pay - Price sensitivity and value perception
- Feature Preferences - Which benefits matter most to customers
- Brand Appeal - Brand name and packaging preferences
Step 3: Request and Test Patch Samples
Sample Development Process
Quality patch sample testing requires obtaining actual product samples from your manufacturing partner. Request small batches that represent your final product specifications:
- Small Batch Samples - Order 50-100 units for initial testing
- Prototype Variations - Test different ingredient concentrations and adhesive types
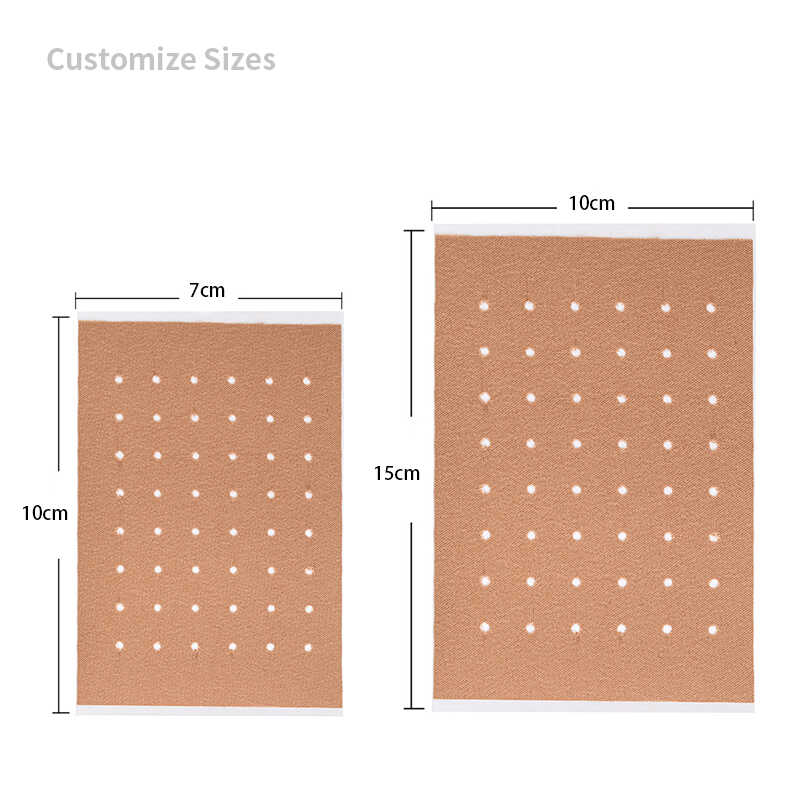
- Different Sizes - Evaluate various patch dimensions for different body areas
- Packaging Options - Assess different packaging formats and materials
Patch Sample Testing Protocol
- Physical Testing - Adhesion strength, durability, and ease of application
- Skin Compatibility - Check for irritation, redness, or allergic reactions
- Efficacy Assessment - Evaluate pain relief duration and intensity
- Comfort Testing - Assess wearability during extended use
- User Experience - Gather feedback on application and removal process
Step 4: Analyze Validation Results
After completing your transdermal patch market test and patch sample testing, analyze the data systematically:
- Quantitative Analysis - Calculate percentages for survey responses and purchase intent
- Qualitative Feedback - Identify recurring themes in customer comments
- Formulation Performance - Compare results across different sample variations
- Cost-Benefit Analysis - Determine if the product can be produced profitably at target price points
Step 5: Make Go/No-Go Decision
Based on validation results, make an informed decision:
- Proceed to Production - If validation shows strong market demand and positive sample feedback
- Iterate and Retest - If issues were identified but are fixable through formulation adjustments
- Pivot or Abandon - If validation reveals fundamental market or product problems
Product Specifications
| Validation Phase | Timeline | Investment |
|---|---|---|
| Concept Definition | 1-2 weeks | Minimal (internal resources) |
| Market Research | 2-4 weeks | $500 - $2,000 |
| Sample Development | 2-4 weeks | $200 - $1,000 |
| Sample Testing | 2-4 weeks | $500 - $3,000 |
| Analysis & Decision | 1-2 weeks | Minimal (internal resources) |
| Total Validation | 8-16 weeks | $1,200 - $6,000 |
Why Choose KONGDY Medical for Your Validation Journey
KONGDY Medical supports your patch product validation process with comprehensive OEM/ODM services. Here is why partnering with us ensures successful product development:
- 35+ Years of Industry Experience - Extensive expertise in transdermal patch formulation and manufacturing
- Small Batch Sample Production - Flexible manufacturing allows cost-effective sample development for validation
- ISO 13485 Certified Quality - Our ISO 13485 certified facility ensures all samples meet international quality standards
- Full R&D Support - Our laboratory team assists with formulation optimization based on your validation feedback
- Competitive Pricing - Efficient production processes provide cost-effective solutions for both samples and full production
- Comprehensive Documentation - Complete technical files and compliance documentation for market registration
Conclusion
Patch product validation is an essential investment that protects your business from costly mistakes and ensures market success. By following a systematic approach to transdermal patch market test and patch sample testing, you can make data-driven decisions about your product concept before committing to large-scale production.
KONGDY Medical stands ready to support your validation journey with expert guidance, quality sample production, and seamless transition to commercial manufacturing. Our comprehensive services ensure that your validated product concept becomes a market-ready reality.
Contact KONGDY Medical today for a free consultation!





