What Are the Common Mistakes When Sourcing a Patch Manufacturer?
Introduction
Selecting the right patch manufacturer is one of the most critical decisions for businesses entering the transdermal patch market. However, many companies fall into common traps during the supplier evaluation process, leading to quality issues, production delays, and financial losses. Understanding these patch sourcing mistakes and OEM supplier risks is essential for making informed decisions and protecting your brand reputation.
Why Choosing the Right Manufacturer Matters
The quality of your transdermal patches directly impacts customer satisfaction, brand credibility, and ultimately your bottom line. Partnering with an inexperienced or unreliable OEM patch manufacturer can result in costly recalls, regulatory compliance issues, and permanent damage to your brand reputation. Therefore, avoiding common patch sourcing mistakes is crucial for long-term business success.
Many businesses underestimate the complexity of transdermal patch manufacturing. Unlike simple consumer products, patches require precise formulation development, strict quality control, and compliance with various international regulations. The consequences of choosing the wrong supplier extend far beyond initial production costs.
Common Patch Sourcing Mistakes to Avoid
Mistake 1: Focusing Only on Price
One of the most prevalent patch sourcing mistakes is selecting a manufacturer based solely on the lowest price. While cost considerations are important, extremely low pricing often indicates compromised quality in one or more areas:
- Substandard Raw Materials - Cheaper ingredients may not meet pharmaceutical-grade requirements
- Inadequate Quality Control - Reduced testing protocols increase the risk of defective products
- Limited Regulatory Compliance - Cost-cutting measures may bypass essential certifications and documentation
- Hidden Costs - Initial savings often lead to expensive problems later, including recalls and rework
A reputable ISO 13485 certified manufacturer may have higher upfront costs but provides better value through consistent quality and reduced risk.
Mistake 2: Not Verifying Manufacturing Certifications
Failing to verify a manufacturer credentials is a critical error that exposes your business to significant OEM supplier risks. Always confirm:
- ISO 13485 Certification - Essential for medical device quality management
- CE Marking Capability - Required for European market access
- FDA Registration - Necessary for selling in the United States
- Production Facility Audit - Physical inspection of manufacturing conditions
- Third-Party Testing Records - Independent verification of product quality
Mistake 3: Ignoring Communication and Transparency
Poor communication is a major warning sign that often leads to patch sourcing mistakes. OEM supplier risks increase significantly when manufacturers:
- Are Unresponsive - Delayed responses indicate potential capacity or priority issues
- Lack Technical Expertise - Cannot explain formulation details or quality processes
- Hide Production Issues - Fail to proactively communicate problems or delays
- Provide Vague Documentation - Cannot supply clear certificates of analysis or batch records
Choose a manufacturer that values transparent communication and provides regular updates throughout the production process.
Mistake 4: Overlooking Minimum Order Quantities and Flexibility
Many businesses make patch sourcing mistakes by not considering their actual business needs. Understanding MOQ requirements helps avoid:
- Excessive Inventory Costs - Tying up capital in slow-moving stock
- Cash Flow Problems - Large upfront orders strain financial resources
- Product Obsolescence - Market changes make large batches outdated
- Storage Challenges - Finding adequate warehouse space for bulk orders
Partner with manufacturers who offer flexible production volumes to match your business growth and market demand.
Mistake 5: Neglecting Quality Assurance Protocols
Another common patch sourcing mistake is assuming that certifications alone guarantee quality. OEM supplier risks related to quality assurance include:
- Inconsistent Batch Quality - Variation between production runs
- Contamination Issues - Cross-contamination during manufacturing
- Incorrect Ingredient Ratios - Formulation errors affecting efficacy
- Packaging Defects - Seal integrity or labeling errors
Request detailed quality documentation including batch testing results, stability studies, and certificates of analysis before committing to a supplier.
How to Evaluate Patch Manufacturers Properly
Avoiding patch sourcing mistakes requires a systematic approach to supplier evaluation. Follow this comprehensive checklist:
- Request Facility Tours - Visit manufacturing plants to observe production conditions firsthand
- Review Sample Products - Test samples for quality, adhesion, and skin compatibility
- Check References - Contact existing clients to verify supplier reliability
- Audit Quality Systems - Review quality manuals, SOPs, and testing protocols
- Evaluate Technical Capabilities - Assess R&D facilities and formulation expertise
- Verify Supply Chain - Understand raw material sourcing and backup suppliers
OEM Supplier Risks of Choosing Wrong
The OEM supplier risks of partnering with the wrong manufacturer extend beyond initial financial losses:
- Regulatory Penalties - Non-compliant products may face market withdrawal and fines
- Brand Reputation Damage - Quality issues erode customer trust and loyalty
- Legal Liabilities - Product liability claims from injured consumers
- Supply Chain Disruption - Production delays impact sales and market timing
- Competitive Disadvantage - Inferior products fail against better-quality competitors
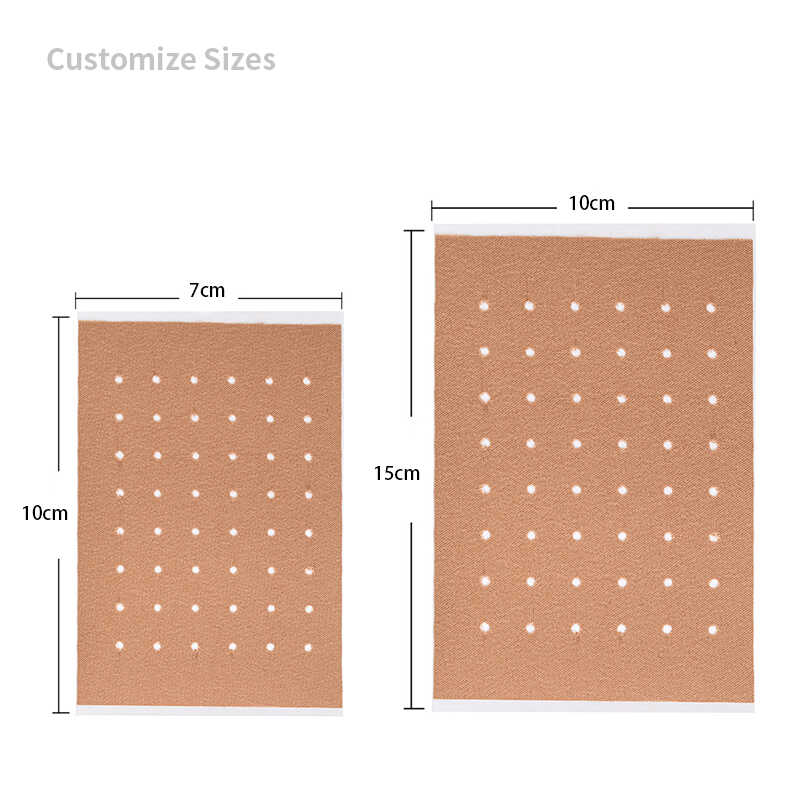
Product Specifications
| Evaluation Criteria | What to Verify |
|---|---|
| Certifications | ISO 13485, CE, FDA registered |
| Production Capacity | Monthly output, equipment, workforce |
| Quality Control | Testing procedures, equipment, documentation |
| Experience | Years in business, client portfolio, product range |
| Communication | Response time, technical support, transparency |
| Pricing Structure | Unit costs, MOQ requirements, payment terms |
| Lead Time | Standard production time, rush options, logistics |
| Scalability | Ability to grow with your business needs |
Why Choose KONGDY Medical as Your Manufacturing Partner
KONGDY Medical eliminates common patch sourcing mistakes by providing reliable, high-quality manufacturing services. Here is why partnering with us minimizes OEM supplier risks:
- 35+ Years of Proven Experience - Extensive track record in transdermal patch manufacturing with consistent quality standards
- ISO 13485 Certified Facility - Meeting the highest international standards for medical device quality management
- Transparent Communication - Dedicated account managers provide regular updates throughout the production process
- Flexible Production Volumes - Scalable manufacturing capacity from small batches to commercial-scale orders
- Comprehensive Quality Assurance - Rigorous testing at every stage from raw materials to finished products
- Complete Regulatory Support - Documentation and compliance assistance for multiple market registrations
Conclusion
Avoiding patch sourcing mistakes requires careful evaluation, due diligence, and a focus on long-term partnership value rather than short-term cost savings. By understanding the common OEM supplier risks and following proper evaluation procedures, you can select a manufacturer that supports your business growth and protects your brand reputation.
KONGDY Medical stands as a reliable partner for businesses seeking quality-conscious patch manufacturing services. Our commitment to quality, transparency, and customer success makes us the ideal choice for brands that refuse to compromise on product excellence.
Contact KONGDY Medical today for a free consultation!





