The Complete Guide to Transdermal Patch Regulatory Requirements
Introduction
Navigating transdermal patch regulatory requirements is essential for any business entering the pharmaceutical or healthcare patch market. Understanding compliance standards, certification processes, and documentation requirements ensures your products meet international safety and quality benchmarks. This comprehensive guide covers the essential regulatory frameworks for transdermal patch manufacturing and market authorization.
Understanding Global Regulatory Frameworks
Transdermal patches are classified as medical devices or pharmaceutical products depending on their intended use and active ingredients. Different markets have distinct regulatory pathways that manufacturers must follow:
- United States - FDA regulates transdermal patches as prescription drugs or over-the-counter (OTC) medications
- European Union - CE marking under the Medical Device Regulation (MDR 2017/745) is required
- China - NMPA (National Medical Products Administration) oversees patch product registration
- Japan - PMDA (Pharmaceuticals and Medical Devices Agency) handles regulatory approvals
- Other Markets - Each country has specific requirements for import and sale
Partnering with an experienced OEM patch manufacturer familiar with these requirements simplifies the compliance process significantly.
Key Quality Management Certifications
ISO 13485 Certification
ISO 13485 is the gold standard for medical device quality management systems. Our ISO 13485 certified facility demonstrates compliance with:
- Quality Management System (QMS) - Comprehensive procedures for design, development, production, and distribution
- Risk Management - Systematic identification and mitigation of product risks
- Document Control - Proper management of quality records and documentation
- Traceability - Complete batch tracking from raw materials to finished products
Other Essential Certifications
- CE Marking - Required for European market access, demonstrating safety and performance compliance
- FDA Registration - Establishes your manufacturing facility with the U.S. Food and Drug Administration
- GMP Compliance - Good Manufacturing Practice standards ensure consistent product quality
- ISO 22716 - Guidelines for cosmetics manufacturing, relevant for wellness patches
Documentation Requirements
Meeting transdermal patch regulatory requirements demands comprehensive documentation throughout the product lifecycle:
- Technical File - Complete product specifications, design history, and risk analysis
- Design History File (DHF) - Records of all design and development activities
- Device Master Record (DMR) - Detailed specifications for manufacturing and quality control
- Batch Production Records - Documentation of each production batch including materials and processes
- Certificates of Analysis (CoA) - Test results confirming product meets specifications
- Stability Study Data - Evidence of product shelf life under specified storage conditions
Product Testing Requirements
Transdermal patches must undergo rigorous testing to demonstrate safety and efficacy:
- Physical Testing - Adhesion strength, tensile properties, and dimensional accuracy
- Chemical Testing - Active ingredient content, impurity profiling, and dissolution rates
- Microbiological Testing - Total viable count, sterility, and preservation efficacy
- Skin Sensitization Testing - Assessment of potential allergic reactions
- Stability Testing - Long-term and accelerated aging studies
- Biocompatibility Testing - Evaluation of skin contact safety
Labeling and Packaging Compliance
Regulatory requirements extend to product labeling and packaging:
- Ingredient Declaration - Complete listing of active and inactive ingredients
- Usage Instructions - Clear directions for application, duration, and precautions
- Storage Conditions - Proper handling and storage guidelines
- Batch Number and Expiry Date - Traceability and shelf life information
- Regulatory Symbols - CE mark, FDA registration number, or other required symbols
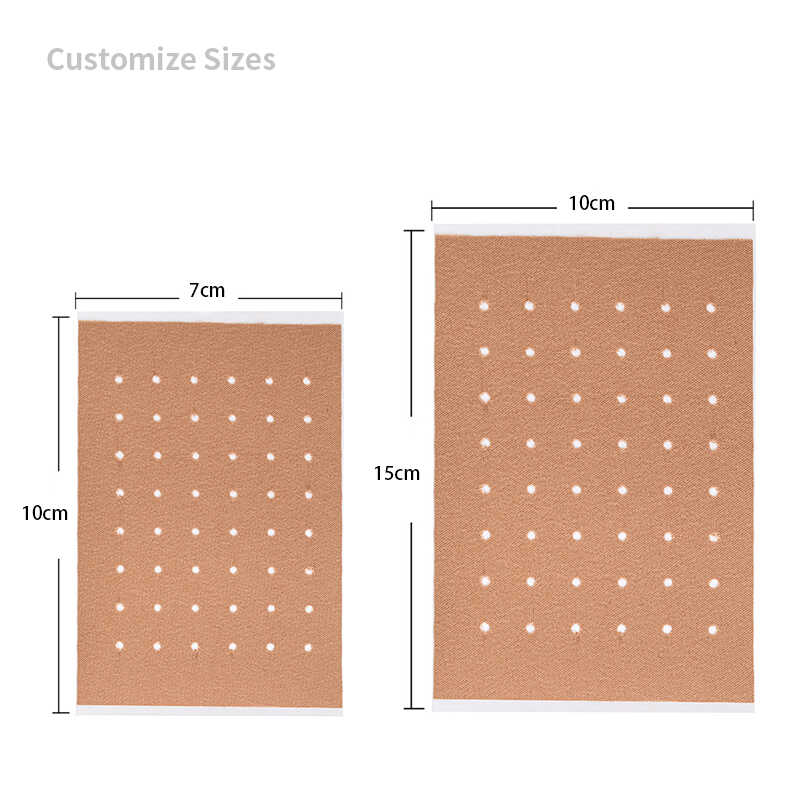
Product Specifications
| Regulatory Aspect | Requirements |
|---|---|
| Quality Certification | ISO 13485:2016 |
| Market Access | CE Marking, FDA Registered |
| Testing Standards | ISO 10993 (Biocompatibility) |
| Documentation | Technical File, DHF, DMR, Batch Records |
| Stability Studies | ICH Q1A compliant protocols |
| Shelf Life | 24 months (standard), longer with validation |
| Manufacturing Records | Complete traceability and CoA |
| Audit Support | Facility audits, documentation review |
Why KONGDY Medical Ensures Regulatory Compliance
KONGDY Medical provides comprehensive regulatory support to simplify your compliance journey. Here is why our regulatory expertise sets us apart:
- 35+ Years of Regulatory Experience - Deep understanding of global regulatory frameworks and compliance requirements
- ISO 13485 Certified Facility - Meeting the highest international standards for medical device quality management
- Complete Documentation Package - Comprehensive technical files prepared according to regulatory specifications
- Full Testing Support - In-house and partner laboratory capabilities for all required testing
- Regulatory Consultation - Expert guidance on market-specific requirements and registration processes
- Ongoing Compliance Monitoring - Continuous updates to ensure continued adherence to changing regulations
Conclusion
Understanding and meeting transdermal patch regulatory requirements is fundamental to successful market entry. By partnering with a manufacturer that prioritizes compliance and maintains rigorous quality standards, you can navigate the complex regulatory landscape with confidence.
KONGDY Medical stands ready to support your regulatory compliance journey with our ISO 13485 certified facility, comprehensive documentation, and expert consultation services. Our commitment to quality ensures that your products meet all necessary requirements for market authorization worldwide.
Contact KONGDY Medical today for a free consultation!





