What Ingredients Are Commonly Used in Transdermal Patches?
Introduction
Transdermal patches are innovative drug delivery systems designed to deliver medications through the skin directly into the bloodstream. Understanding transdermal patch ingredients and patch formulation is essential for manufacturers seeking to develop effective therapeutic products. These patches offer controlled release, improved bioavailability, and non-invasive administration compared to traditional oral medications.
What Are Transdermal Patches?
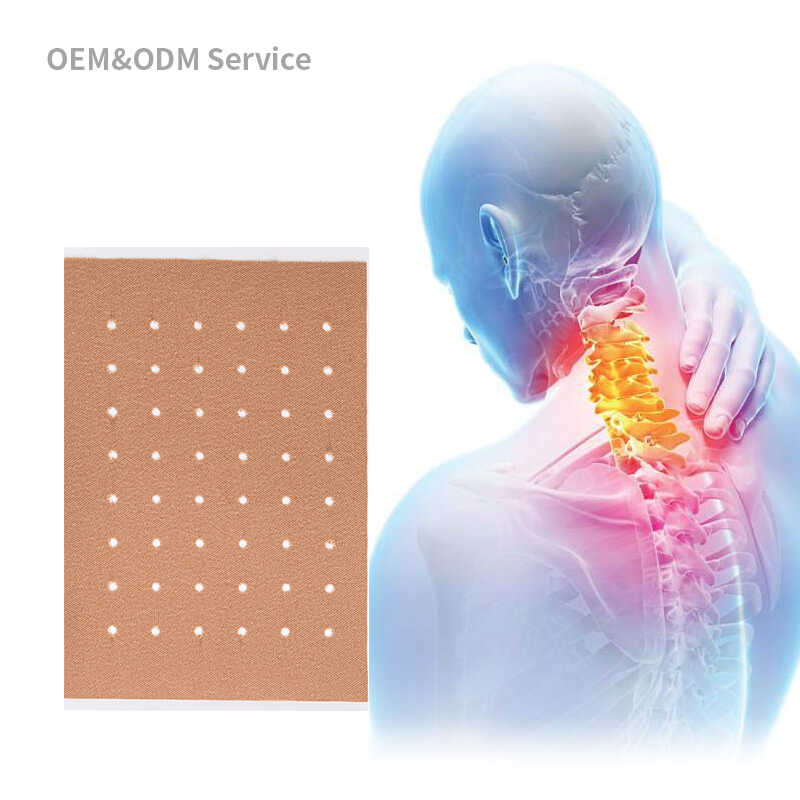
Transdermal patches are adhesive devices containing medication that adhere to the skin surface and deliver drugs systematically. Originally developed for motion sickness and hormone replacement therapy, transdermal technology now addresses conditions ranging from chronic pain and nicotine dependence to cardiovascular diseases and neurological disorders. The effectiveness of these patches depends significantly on the careful selection and formulation of transdermal patch ingredients.
Modern transdermal patches consist of multiple layers, each serving a specific function in the delivery system. The backing layer provides structural support and protects the formulation from external factors. The drug reservoir holds the active pharmaceutical ingredients along with excipients that enhance drug stability and permeability. The adhesive layer ensures proper skin contact throughout the wear period, while the release liner is removed before application.
Key Transdermal Patch Ingredients
The formulation of effective transdermal patches requires a precise combination of pharmaceutical-grade ingredients. Each component plays a critical role in ensuring drug stability, skin penetration, and therapeutic efficacy.
Active Pharmaceutical Ingredients (APIs)
The active pharmaceutical ingredient is the core component that provides therapeutic effect. Common APIs in transdermal formulations include:
- Menthol (3-10%) - Provides cooling sensation and enhances local blood circulation
- Camphor (2-8%) - Offers mild analgesic effects and improves skin penetration
- Capsaicin (0.025-0.075%) - Delivers long-lasting pain relief for neuropathic and musculoskeletal conditions
- Methyl Salicylate (10-30%) - Provides anti-inflammatory benefits for joint and muscle pain
- Lidocaine (2-5%) - Acts as a local anesthetic for rapid pain relief
- Diclofenac Diethylamine (1-2%) - Delivers potent anti-inflammatory action for arthritis relief
Adhesive Polymers
Adhesive components ensure the patch remains securely attached to the skin throughout the application period:
- Polyisobutylene (PIB) - Provides excellent adhesion and moisture resistance
- Acrylic Polymers - Offer versatile adhesion properties and drug compatibility
- Silicone Adhesives - Deliver gentle adhesion suitable for sensitive skin
- Hydrocolloids - Absorb exudate while maintaining moist wound healing environment
Penetration Enhancers
These ingredients improve drug absorption through the stratum corneum:
- Oleic Acid (1-10%) - Disrupts skin barrier and increases lipid fluidity
- Propylene Glycol (5-20%) - Acts as solvent and penetration accelerator
- Transcutol (2-10%) - Enhances drug solubility and skin permeation
- Azone (1-5%) - Provides long-lasting penetration enhancement
Excipients and Fillers
- Dibutyl Phthalate (plasticizer) - Improves flexibility of polymer matrix
- Ethyl Cellulose - Provides film-forming properties
- Silicon Dioxide - Acts as flow agent and desiccant
- Antioxidants (BHT, Vitamin E) - Prevent oxidative degradation of active ingredients
OEM Manufacturing Process for Transdermal Patches
1. Formula Development
The OEM manufacturing process begins with extensive research and development. Our R&D team conducts solubility studies, stability testing, and permeability assays to optimize the patch formulation. We evaluate different combinations of transdermal patch ingredients to achieve the desired release profile and therapeutic effect.
During formula development, we consider factors such as drug physicochemical properties, target tissue depth, required dosing frequency, and patient compliance. Our laboratory employs advanced technologies including Franz diffusion cells for skin permeation testing and high-performance liquid chromatography for content uniformity analysis.
2. Raw Material Preparation
All pharmaceutical-grade transdermal patch ingredients undergo rigorous quality control inspection upon receipt. Raw materials are tested for identity, purity, particle size distribution, and microbial contamination. Only materials meeting our strict specifications proceed to the manufacturing stage.
Ingredients are precisely weighed according to validated formulas using calibrated analytical balances. The weighing process follows GMP protocols with complete documentation and traceability. Temperature and humidity conditions are controlled throughout material handling to prevent moisture absorption or degradation.
3. Production Manufacturing
Our ISO 13485 certified manufacturing facility operates advanced production lines for transdermal patch fabrication. The production process includes:
- Mixing and Blending - Active ingredients and excipients are homogeneously mixed using industrial mixers
- Coating Application - The formulation is coated onto backing materials using precision coating machines
- Drying and Curing - Solvents are removed through controlled drying tunnels
- Lamination - Multiple layers are bonded together under controlled pressure
- Slitting and Cutting - Large rolls are cut into individual patches of specified dimensions
- Packaging - Patches are sealed in protective pouches to maintain stability
4. Quality Control
Every batch of transdermal patches undergoes comprehensive quality testing before release. Our quality assurance team performs:
- Physical Testing - Weight uniformity, thickness, and adhesion strength measurement
- Chemical Analysis - HPLC testing for API content and impurity profiling
- Performance Testing - In vitro drug release studies using dissolution apparatus
- Stability Studies - Accelerated aging tests under controlled temperature and humidity
- Microbiological Testing - Total viable count, yeast and mold, and pathogen screening
Product Specifications
| Parameter | Specification |
|---|---|
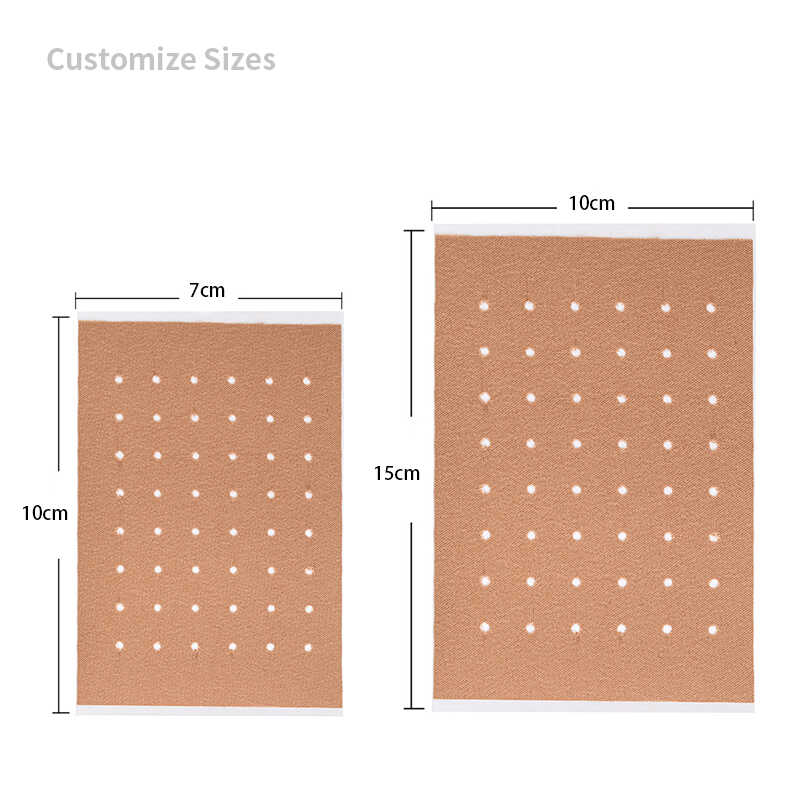
| Patch Size | 7cm x 10cm, 10cm x 14cm, 12cm x 18cm (customizable) |
| Patch Thickness | 0.3mm - 1.0mm |
| Active Ingredient Content | Menthol 3-10%, Camphor 2-8%, Capsaicin 0.025-0.075% |
| Wearing Time | 8-12 hours per application |
| Shelf Life | 24 months in original packaging |
| Storage Conditions | Temperature 15-30°C, Relative Humidity below 60% |
| Packaging Options | Individual sealed pouches, multi-pack boxes |
| Certifications | ISO 13485, CE, FDA registered |
Why Choose KONGDY Medical?
As a leading transdermal patch manufacturer, KONGDY Medical offers comprehensive OEM/ODM services for pharmaceutical and healthcare companies worldwide. Here are the key advantages of partnering with us:
- 35+ Years of Industry Experience - Our extensive expertise in transdermal patch formulation development ensures optimal results for your specific therapeutic requirements.
- ISO 13485 Certified Facility - We maintain rigorous quality management systems that meet international medical device standards, guaranteeing product safety and efficacy.
- Advanced R&D Capabilities - Our state-of-the-art laboratory features cutting-edge equipment for transdermal patch ingredients analysis, formulation optimization, and performance testing.
- Competitive Pricing - Our efficient production processes and economies of scale allow us to offer cost-effective solutions without compromising on quality.
- Full OEM/ODM Support - From concept development to final packaging, we provide complete private label manufacturing services tailored to your brand requirements.
- Fast and Reliable Delivery - Our streamlined logistics network ensures timely delivery of your orders, meeting your market launch schedules consistently.
Conclusion
Understanding the science behind transdermal patch ingredients and patch formulation is crucial for developing effective transdermal delivery systems. The precise combination of active pharmaceutical ingredients, adhesive polymers, penetration enhancers, and excipients determines the therapeutic performance and patient acceptance of the final product.
At KONGDY Medical, we possess the expertise, technology, and quality systems necessary to transform your transdermal patch concepts into market-ready products. Our commitment to excellence in every aspect of patch formulation and manufacturing makes us the ideal partner for your transdermal delivery needs.
Contact KONGDY Medical today for a free consultation!





