How Long Does It Take to Develop a Custom Transdermal Patch?
Introduction
The transdermal patch market is experiencing unprecedented growth, with entrepreneurs and healthcare brands increasingly seeking custom patch OEM solutions. Understanding the patch development timeline is crucial for planning your product launch and market entry strategy.
From initial formulation development to final production, the custom patch OEM process typically spans 3-6 months depending on complexity. This comprehensive guide breaks down each phase of transdermal patch development with realistic timelines.
Understanding the Transdermal Patch Development Timeline
What Affects Patch Development Time?
Several factors influence the overall patch development timeline:
- Formulation Complexity: Simple over-the-counter formulations require less R&D time than pharmaceutical-grade products
- Regulatory Requirements: Markets requiring extensive documentation extend timelines
- Testing Requirements: Additional stability or clinical testing adds weeks to the schedule
- Material Sourcing: Specialized ingredients may require longer procurement periods
- Customization Level: Full custom formulations take longer than private label modifications
Average Development Timeline Overview
For a standard private label patch OEM project, expect the following timeline:
- Phase 1 - Consultation & Planning: 1-2 weeks
- Phase 2 - Formulation Development: 4-8 weeks
- Phase 3 - Prototype & Testing: 4-6 weeks
- Phase 4 - Regulatory & Documentation: 2-4 weeks
- Phase 5 - Production Setup: 2-3 weeks
- Phase 6 - Initial Production: 3-4 weeks
Total Average Timeline: 12-27 weeks (3-6 months)
Detailed Breakdown of Each Development Phase
Phase 1: Consultation & Planning (Week 1-2)
The custom patch OEM process begins with comprehensive consultation. During this phase:
- Define product specifications and target market
- Select active pharmaceutical ingredients (APIs) and excipients
- Discuss packaging requirements and branding guidelines
- Establish minimum order quantity (MOQ) and pricing structure
- Review regulatory requirements for target markets
Timeline: 1-2 weeks
Phase 2: Formulation Development (Week 3-10)
This is the most critical phase of the patch development timeline. Our R&D team works on:
- API Selection: Identifying optimal active ingredients based on solubility and permeability
- Matrix Development: Creating the adhesive matrix formulation
- Permeation Enhancement: Adding enhancers to improve drug absorption through skin
- Backing Material Selection: Choosing appropriate backing layers for comfort and durability
- Prototype Formulations: Preparing multiple formulation variants for testing
Typical Ingredients Used:
- Menthol (3-10%): Provides cooling sensation
- Camphor (1-5%): Analgesic properties
- Capsaicin (0.025-0.1%): Natural pain relief
- Methyl Salicylate (10-30%): Anti-inflammatory effects
- Lidocaine (2-5%): Local anesthetic
Timeline: 4-8 weeks
Phase 3: Prototype & Testing (Week 11-16)
Once formulations are developed, rigorous testing ensures product efficacy and safety:
- Content Uniformity Testing: Verifying consistent drug distribution across patches
- Adhesion Testing: Ensuring patches stick properly for required duration
- Skin Compatibility Testing: Checking for irritation or allergic reactions
- In Vitro Permeation Studies: Measuring drug release rates
- Stability Testing: Assessing shelf-life under various conditions
Timeline: 4-6 weeks
Phase 4: Regulatory & Documentation (Week 17-20)
The ISO 13485 certified regulatory team prepares comprehensive documentation:
- Certificate of Analysis (CoA): Detailed product specifications and test results
- Material Safety Data Sheets (MSDS): Handling and safety information
- Technical Files: Complete manufacturing documentation
- Regulatory Submissions: Documentation for target market approvals
- Quality Agreements: Contracts defining quality responsibilities
Timeline: 2-4 weeks
Phase 5: Production Setup (Week 21-23)
Before mass production begins, production lines are configured:
- Equipment Calibration: Setting up coating and lamination machines
- Tooling Preparation: Creating custom dies and molds for packaging
- Material Ordering: Sourcing bulk raw materials and packaging supplies
- Quality Control Procedures: Implementing inspection protocols
- Pilot Run: Small batch production to validate processes
Timeline: 2-3 weeks
Phase 6: Initial Production & Delivery (Week 24-27)
The final phase of the patch development timeline includes:
- Mass Production: Full-scale manufacturing runs
- Quality Inspection: Random sampling and testing of finished products
- Packaging: Individual and retail packaging with your branding
- Quality Release: Final approval based on quality standards
- Shipping & Logistics: Export preparation and delivery coordination
Timeline: 3-4 weeks

Timeline Variations: Standard vs. Express Development
Standard Development Timeline
The complete custom patch OEM process with full testing and documentation typically takes 4-6 months. This timeline includes comprehensive quality assurance and is recommended for pharmaceutical-grade products or markets with strict regulatory requirements.
Express Development Timeline
For established formulations with minor customizations, the private label development timeline can be compressed to 10-14 weeks. This option works best for brands using proven formulations with custom branding only.
Timeline Comparison Table
| Development Type | Timeline | Best For |
|---|---|---|
| Express | 10-14 weeks | Private label, proven formulas |
| Standard | 16-20 weeks | Custom formulations, new markets |
| Comprehensive | 24-27 weeks | Pharmaceutical-grade, FDA submissions |
How to Accelerate Your Patch Development Timeline
Want to shorten your patch development timeline? Consider these strategies:
- Start with Proven Formulations: Using existing KONGDY Medical formulations reduces R&D time significantly
- Prepare Documentation Early: Start gathering regulatory documents during formulation development
- Parallel Processing: Conduct testing and documentation simultaneously where possible
- Clear Communication: Respond promptly to feedback and approval requests
- Early Material Sourcing: Pre-order critical materials to avoid production delays
Product Specifications for Common Patch Types
| Specification | Pain Relief Patch | Cooling Gel Patch |
|---|---|---|
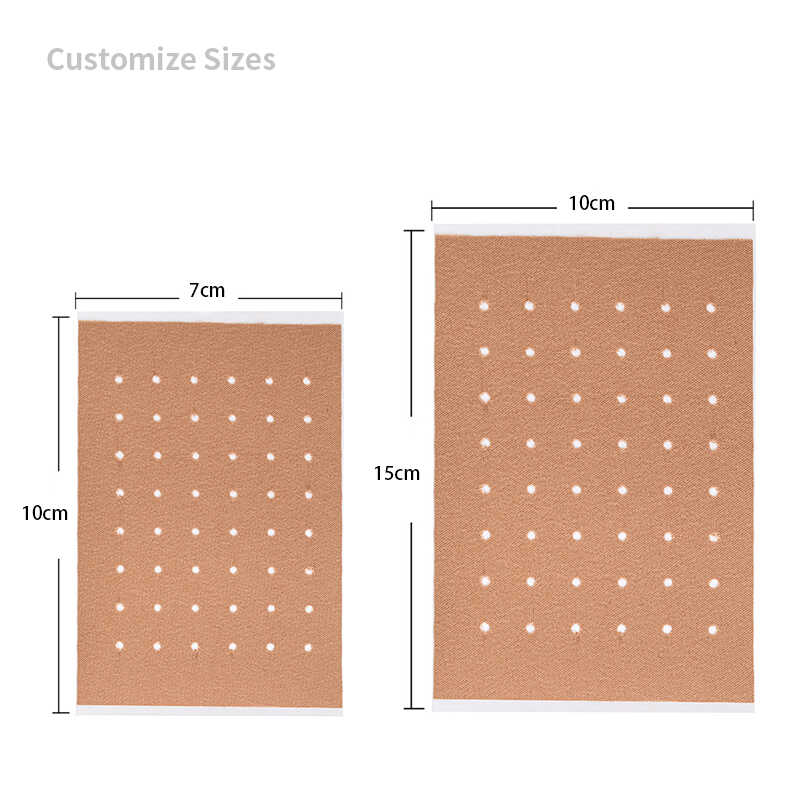
| Size | 7cm x 10cm, 10cm x 14cm | 5cm x 12cm, 8cm x 15cm |
| Duration | 8-12 hours | 4-8 hours |
| Shelf Life | 24 months | 18-24 months |
| MOQ | 5,000 pieces | 5,000 pieces |
Why Partner with KONGDY Medical for Patch Development
1. Extensive Experience
With over 35 years in transdermal patch manufacturing, KONGDY Medical has developed hundreds of custom formulations for clients worldwide. Our experienced team understands the nuances of transdermal drug delivery.
2. State-of-the-Art R&D Facilities
Our dedicated research center features advanced formulation development labs, permeation testing equipment, and pilot production lines. This enables us to accelerate the patch development timeline while maintaining rigorous quality standards.
3. Comprehensive Testing Capabilities
From in-house content uniformity testing to third-party stability studies, we offer comprehensive quality assurance. Every batch undergoes strict inspection before release.
4. Flexible MOQ Options
Starting at just 5,000 pieces, our OEM manufacturing services are accessible for businesses of all sizes. Volume discounts apply for orders over 10,000 pieces.
5. Global Regulatory Expertise
Our regulatory affairs team has successfully registered products in 60+ countries. We handle documentation preparation, submission tracking, and compliance monitoring throughout the custom patch OEM process.
6. Reliable Delivery
Standard production lead time is 3-4 weeks after sample approval. Express options are available for urgent orders. We guarantee on-time delivery with full tracking visibility.
Conclusion
Understanding the patch development timeline is essential for successful product launches. While the complete custom patch OEM process typically takes 3-6 months, working with an experienced manufacturer like KONGDY Medical can significantly streamline your timeline.
Our team of 25+ pharmaceutical scientists, state-of-the-art facilities, and proven track record ensure your transdermal patch reaches market efficiently and profitably.
Ready to start your patch development project?
WhatsApp: +86 18939260319
Email: kongdypatch@gmail.com
Website: https://www.kongdymedical.com





