What Are the Common Mistakes When Sourcing a Patch Manufacturer?
Introduction
Choosing the right patch manufacturer is one of the most critical decisions for your private label business. However, many buyers fall into common traps that lead to quality issues, production delays, financial losses, and damaged brand reputation. This comprehensive guide explores the most costly patch sourcing mistakes and OEM supplier risks, helping you make informed decisions when selecting your manufacturing partner.
The medical patch industry has grown exponentially, with numerous suppliers claiming to offer premium OEM services. Distinguishing between reliable partners and substandard manufacturers requires careful evaluation. By understanding these common mistakes, you can protect your investment and establish a successful private label business.
Understanding Patch Sourcing Mistakes in the OEM Industry
The Importance of Proper Supplier Evaluation
Rushing through the supplier selection process is perhaps the most dangerous patch sourcing mistake. Many buyers, eager to launch products quickly, skip essential due diligence steps. This approach often leads to partnering with manufacturers who cannot deliver consistent quality or meet production timelines.
A thorough evaluation should include facility inspections, quality certifications verification, sample testing, and financial stability assessment. The initial time investment in proper vetting saves significant costs and headaches later.
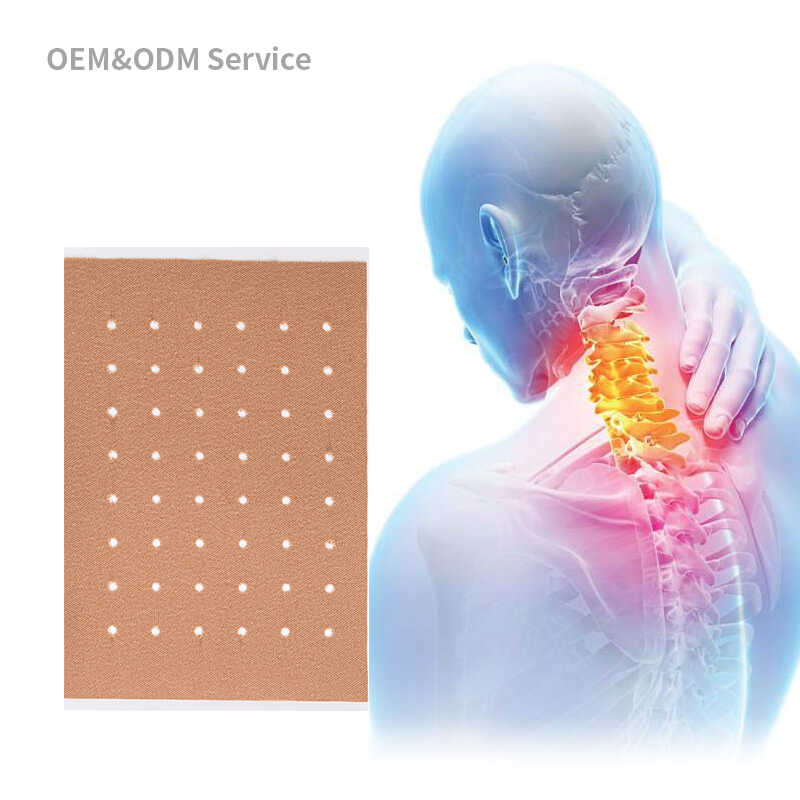
Critical Patch Sourcing Mistakes to Avoid
Mistake 1: Ignoring Quality Certifications
One of the most prevalent patch sourcing mistakes is overlooking essential quality certifications. Without proper certifications, you risk:
- Regulatory Non-Compliance: Products may be seized or banned in target markets
- Product Liability Issues: Liability claims can devastate your business
- Reputation Damage: Poor quality products harm your brand image permanently
Essential certifications your OEM supplier risks assessment should include:
| Certification | Purpose | Market Requirement |
|---|---|---|
| ISO 13485 | Medical device quality management | Global compliance |
| FDA Registration | US market access | Mandatory for USA |
| CE Marking | European market access | Required for EU |
| GMP Certification | Manufacturing standards | Many markets require |
| CFDA | China market approval | Required for China |
KONGDY Medical maintains all major international certifications, providing our private label partners with full regulatory compliance and market access confidence.
Mistake 2: Prioritizing Price Over Value
Focusing solely on lowest price is a critical OEM supplier risk that often backfires spectacularly. While competitive pricing matters, extremely low quotes typically indicate:
- Inferior Raw Materials: Cheap ingredients compromise product effectiveness and safety
- Substandard Manufacturing: Outdated equipment and unqualified workers
- Hidden Costs: Quality issues, returns, and reorders inflate true costs
- Minimum Quality Standards: Products barely meeting basic requirements
The true cost of cheap manufacturing includes: product recalls, regulatory fines, lost customers, and brand rehabilitation expenses. Quality OEM suppliers like KONGDY Medical offer transparent pricing that reflects genuine manufacturing value.
Mistake 3: Skipping Sample Testing
Failing to test samples before committing to large orders represents one of the most costly patch sourcing mistakes. Sample evaluation should include:
Physical Testing:
- Adhesive strength: 8-15 N/25mm for optimal performance
- Skin compatibility: No significant irritation after 24-hour wear
- Backing material durability: No tearing during normal use
- Active ingredient content: HPLC verification of labeled percentages
Performance Testing:
- Wear duration: Products should maintain efficacy for claimed timeframe
- Shelf life stability: Greater than or equal to 95% potency at expiry
- Consistency across batches: Uniform quality in each production run
Packaging Evaluation:
- Seal integrity: No air leakage or contamination
- Label accuracy: Correct information and regulatory compliance
- Consumer usability: Easy application and removal
At KONGDY Medical, we provide comprehensive sample testing services, allowing private label partners to verify all quality parameters before mass production.

OEM Supplier Risks in Communication and Transparency
Mistake 4: Poor Communication Channels
Many patch sourcing mistakes stem from inadequate communication with suppliers. Warning signs include:
- Delayed responses (more than 24-48 hours)
- Vague answers to specific questions
- Reluctance to provide facility documentation
- Language barriers affecting clarity
- Unwillingness to schedule video calls or virtual tours
Effective communication is essential for: order updates, quality issue resolution, regulatory compliance coordination, and long-term partnership success.
Mistake 5: Lack of Production Transparency
Choosing suppliers who cannot provide transparent production processes creates significant OEM supplier risks:
- Unverifiable Production Volume: Cannot confirm manufacturing capacity
- Hidden Subcontracting: Work delegated to unqualified facilities
- Inventory Opacity: No visibility into raw material sourcing
- Quality Control Secrecy: Manufacturing secrets prevent accountability
Reputable manufacturers should welcome transparency through: factory tour opportunities, real-time production updates, detailed batch documentation, and quality certificates for each shipment.
Mistake 6: Ignoring Minimum Order Requirements
Understanding MOQ (Minimum Order Quantity) terms prevents misunderstandings that derail business plans. Common OEM supplier risks related to MOQ include:
| MOQ Issue | Consequence | Solution |
|---|---|---|
| Overly high MOQ | Excessive initial inventory investment | Negotiate phased orders |
| Hidden MOQ for materials | Unexpected costs | Request complete BOM |
| MOQ increases without notice | Business planning disruption | Include MOQ terms in contract |
| MOQ tied to expensive items | Cash flow strain | Negotiate component flexibility |
KONGDY Medical offers flexible MOQ options for private label partners, supporting both startup launches and established brand scaling.
Common Patch Sourcing Mistakes in Contract Negotiations
Mistake 7: Vague Quality Specifications
Entering agreements without detailed quality specifications invites OEM supplier risks. Your contract must include:
Product Specifications Table:
| Parameter | Standard | Tolerance |
|---|---|---|
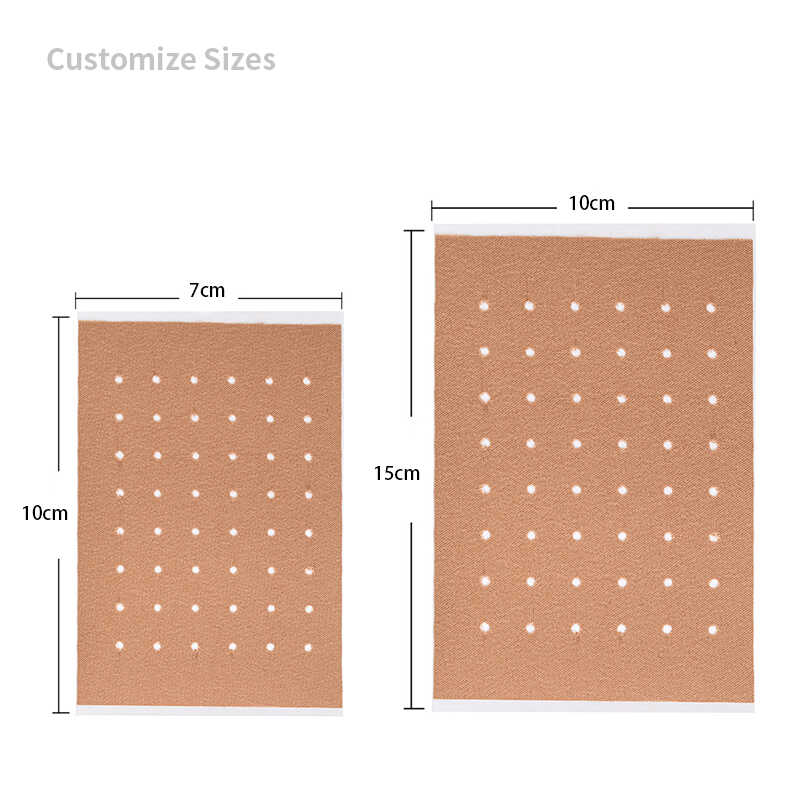
| Patch Size | 7cm x 10cm | Plus/minus 2mm |
| Active Ingredient (Menthol) | 5% | Plus/minus 0.5% |
| Adhesive Strength | 10 N/25mm | 8-15 N/25mm |
| Wear Duration | 8 hours | 6-10 hours claimed |
| Shelf Life | 24 months | Greater than or equal to 95% potency at expiry |
| Packaging | Blister pack | Individual sealed units |
Quality Assurance Terms:
- Defect rate maximum: Less than 1%
- Inspection sampling plan: AQL 1.0
- Quality dispute resolution procedures
- Product liability allocation
Mistake 8: Inadequate Intellectual Property Protection
Protecting your brand identity requires careful contract language. Common patch sourcing mistakes include:
- No Non-Disclosure Agreements: Formulas and designs vulnerable to competitors
- Shared Proprietary Rights: Supplier claims partial ownership of your formulations
- No Exclusivity Clauses: Manufacturer sells identical products to other buyers
- Unclear Branding Rights: Ambiguous ownership of custom designs and packaging
KONGDY Medical implements comprehensive IP protection policies, including strict NDAs, exclusive formulation rights, and complete brand confidentiality.
Mistake 9: Ignoring Payment Terms and Financial Risks
OEM supplier risks extend to financial arrangements. Protect yourself by addressing:
Payment Structure Recommendations:
- 30% deposit upon order confirmation
- 40% payment before production start
- 30% balance upon quality inspection approval
- Never pay 100% upfront to unknown suppliers
Financial Risk Mitigation:
- Verify supplier business license and registration
- Check company financial stability reports
- Request trade references from existing clients
- Use escrow services for large transactions
- Maintain multiple supplier relationships
Logistics and Compliance OEM Supplier Risks
Mistake 10: Underestimating Lead Times and Logistics
Production delays and shipping complications represent patch sourcing mistakes that disrupt supply chains. Consider:
Lead Time Factors:
- Standard production: 15-25 working days
- Custom formulations: Add 10-15 days for development
- Packaging production: Add 5-7 days
- Quality inspection: 3-5 days
- Shipping time: Varies by method and destination
Logistics Risk Management:
- Source multiple shipping providers
- Include buffer time in product launches
- Understand customs procedures for each target market
- Verify supplier has export documentation experience
- Consider warehouse storage options near major markets
Mistake 11: Neglecting Regulatory Compliance Verification
One of the most serious OEM supplier risks involves regulatory non-compliance. Your supplier must demonstrate:
Market-Specific Compliance:
- United States: FDA registration, 21 CFR Part 820 compliance
- European Union: CE marking under MDR 2017/745
- Australia: TGA inclusion if required
- Canada: Health Canada medical device license
- Japan: PMDA registration for specific product classes
Documentation Requirements:
- Technical files and design histories
- Risk analysis documentation
- Clinical evaluation reports
- Post-market surveillance procedures
- Batch production records
KONGDY Medical provides comprehensive regulatory support, including documentation preparation, registration assistance, and compliance verification for all major markets.
Quality Control Patch Sourcing Mistakes
Mistake 12: Assuming All Manufacturers Follow the Same Standards
Quality control patch sourcing mistakes occur when buyers assume uniformity across manufacturers. Reality shows significant variations:
| Quality Aspect | Premium Manufacturer | Budget Manufacturer |
|---|---|---|
| Quality Control Checks | Every batch, multiple stages | Final inspection only |
| Testing Equipment | HPLC, GC, FTIR, dissolution testers | Basic equipment |
| Staff Qualifications | Pharmaceutical trained personnel | General manufacturing |
| Documentation | Complete batch records | Minimal records |
| Rejection Rate | Less than 0.5% | 3-5% accepted defects |
KONGDY Medical implements rigorous quality control at every production stage: incoming material inspection, in-process quality checks, finished product testing, and stability monitoring.
Mistake 13: Not Establishing Clear Defect Standards
Defining acceptable quality levels prevents disputes and protects your business. Essential standards include:
Defect Categories:
| Defect Type | Critical | Major | Minor |
|---|---|---|---|
| Adhesive Failure | Yes | - | - |
| Wrong Labeling | Yes | - | - |
| Color Variation | - | Yes | - |
| Packaging Damage | - | Yes | - |
| Minor Size Variation | - | - | Yes |
Acceptance Criteria:
- AQL 1.0 for critical defects
- AQL 2.5 for major defects
- AQL 4.0 for minor defects
- 100% inspection for critical safety issues
How to Avoid These Patch Sourcing Mistakes
A Comprehensive Supplier Evaluation Checklist
Use this checklist to avoid patch sourcing mistakes:
Phase 1: Initial Screening
- Verify business license and legal registration
- Check years of industry experience (minimum 5 years recommended)
- Review company certifications and accreditations
- Assess production capacity and facilities
- Request client references
Phase 2: Quality Evaluation
- Obtain product samples for testing
- Request quality documentation samples
- Verify testing equipment and procedures
- Review quality control processes
- Assess raw material sourcing practices
Phase 3: Business Assessment
- Evaluate communication responsiveness
- Review pricing structure transparency
- Verify IP protection policies
- Assess financial stability
- Check logistics and shipping capabilities
Phase 4: Contract Negotiation
- Define detailed product specifications
- Establish clear quality standards
- Set reasonable MOQ terms
- Include IP protection clauses
- Define payment terms and schedules
- Establish quality dispute resolution procedures
Why Choose KONGDY Medical to Avoid These Mistakes
Partnering with an experienced manufacturer eliminates most common patch sourcing mistakes:
1. 35+ Years Industry Experience
With over three decades of medical patch manufacturing experience, KONGDY Medical has developed proven processes that eliminate quality issues and production delays. Our track record demonstrates consistent delivery of premium products.
2. Complete Certification Portfolio
We maintain all essential certifications: ISO 13485, FDA registration, CE marking, GMP compliance, and market-specific approvals. Your products meet regulatory requirements worldwide.
3. Rigorous Quality Control
Our multi-stage quality control system ensures every patch meets specifications. We reject any product that does not meet our 99.5% quality standard, protecting your brand reputation.
4. Transparent Operations
KONGDY Medical welcomes facility tours, provides detailed documentation, and maintains open communication. No hidden subcontracting or production surprises.
5. IP Protection Commitment
We implement comprehensive confidentiality policies, sign detailed NDAs, and respect exclusive formulation rights. Your brand identity remains protected.
6. Flexible Partnership Terms
From startup MOQ options to large-scale production, we adapt to your business needs. Our transparent pricing eliminates hidden costs and surprises.
Conclusion
Avoiding patch sourcing mistakes requires careful evaluation, clear communication, and partnership with a reliable manufacturer. By understanding common OEM supplier risks and implementing proper due diligence, you can protect your investment and build a successful private label business.
The consequences of poor supplier selection extend beyond immediate financial losses—damaged reputation, regulatory issues, and lost customer trust can take years to rebuild. Choose a manufacturing partner who demonstrates commitment to quality, transparency, and long-term partnership success.
KONGDY Medical stands ready to help you avoid these costly mistakes. Our decades of experience, comprehensive certifications, and rigorous quality standards provide the foundation for your private label success.
Contact KONGDY Medical today for a free consultation and supplier evaluation assistance!





