OEM Transdermal Patch Manufacturing - Complete Quality Control Guide 2026
Introduction
Quality control in OEM transdermal patch manufacturing determines product safety, efficacy, and market success. This comprehensive guide covers the complete QC process that reputable manufacturers implement.
What is OEM Transdermal Patch Manufacturing?
OEM transdermal patches are drug delivery systems manufactured by specialized factories for brand owners. Key applications include pain relief, fever reduction, smoking cessation, and hormone therapy.
Key Ingredients in Transdermal Patches
Active Ingredients
- Menthol: 1-10% concentration for cooling sensation
- Capsaicin: 0.025-0.1% for warming pain relief
- Camphor: 3-11% for counter-irritant effect
- Methyl Salicylate: 10-30% for anti-inflammatory action
Adhesive Components
- Pressure-sensitive adhesives (acrylic, silicone, rubber-based)
- Matrix formers (polyisobutylene, silicone elastomers)
- Penetration enhancers (terpenes, fatty acids)
Backing Materials
- Non-woven fabrics for breathability
- Polyester films for occlusion
- Foam laminates for cushioning
OEM Manufacturing Process
1. Formula Development
- Active ingredient compatibility testing
- Adhesive compatibility studies
- Skin sensitivity assessment
- Drug release profile optimization
2. Raw Material Preparation
- API weighing and verification (accuracy: ±5%)
- Excipient sourcing from approved suppliers
- Material identity and purity testing
3. Production Manufacturing
| Stage | Process | Controls |
|---|---|---|
| Mixing | Blend APIs with adhesive | Time, speed, temp |
| Coating | Apply to backing | Weight ±10% |
| Lamination | Apply release liner | Pressure, speed |
| Slitting | Cut to sizes | ±0.5mm accuracy |
| Pouching | Individual packaging | Seal integrity |
4. Quality Control Testing

In-Process Controls
- Appearance inspection
- Weight uniformity (CV < 5%)
- Thickness measurement
- Adhesion testing
Finished Product Testing
- Drug content: HPLC analysis (90-110%)
- Drug release: USP dissolution
- Peel force: 1.5-3.0 N/25mm
- Skin sensitivity: Human patch test
Stability Testing
- Accelerated (40°C/75% RH, 6 months)
- Real-time (25°C/60% RH, 24-36 months)
Product Specifications
| Parameter | Specification |
|---|---|
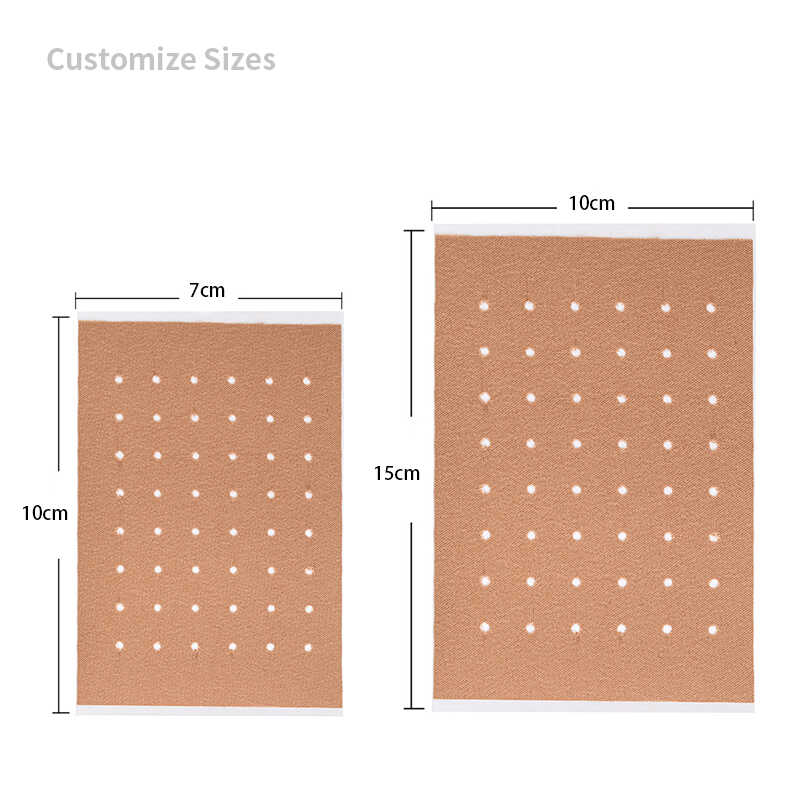
| Patch Sizes | 7x10cm, 10x14cm, custom |
| Active Ingredients | Menthol 1-10%, Capsaicin 0.025-0.1% |
| Wear Duration | 8-12 hours |
| Shelf Life | 24-36 months |
| Packaging | Foil pouch, 5-10 patches/box |
Why Choose KONGDY Medical
KONGDY Medical offers comprehensive OEM manufacturing:
- 35+ years experience: Proven track record in transdermal patch production
- ISO 13485 certified: Medical device quality management system
- R&D capability: Professional formula development team
- Competitive pricing: Cost-effective manufacturing solutions
- OEM/ODM support: Full-service from formula to packaging
- Fast delivery: Efficient production and logistics
Conclusion
Quality control is essential in OEM transdermal patch manufacturing. Partner with KONGDY Medical for reliable, compliant, and cost-effective patch production. Contact KONGDY Medical today for a free consultation!





